Polarity Of Bonds Chart
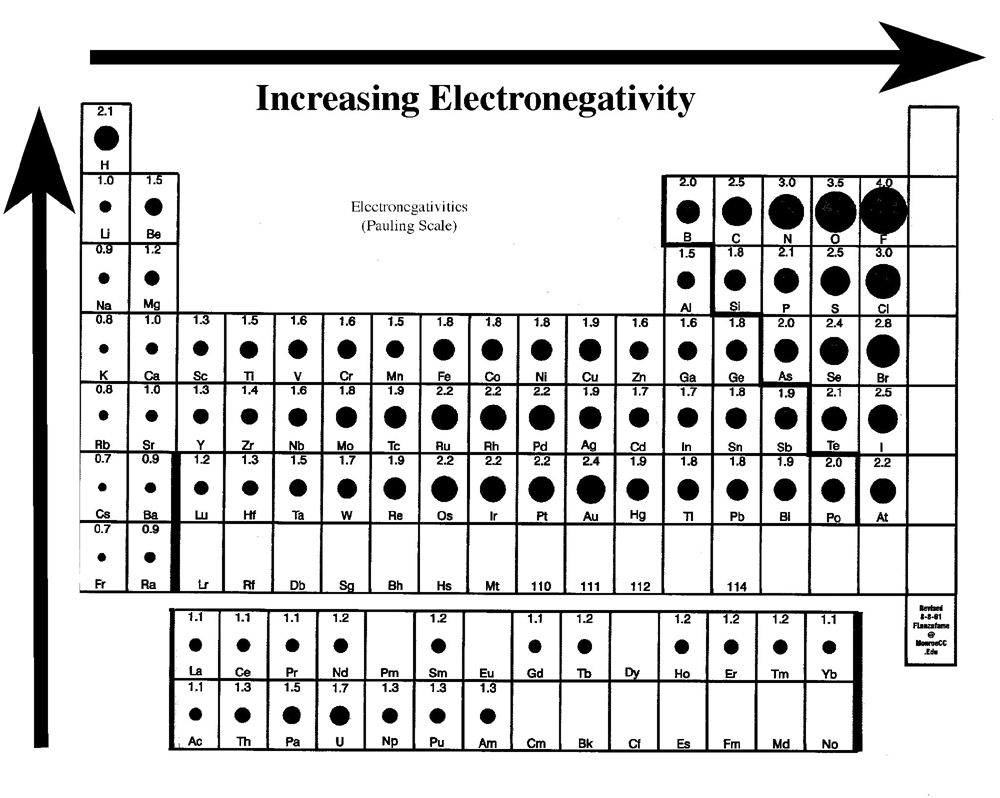
Electronegativity periodic table chart chemistry blank bonds polarity printable covalent elements polar bond worksheet electronegativities sheet determine atomic molecule reference Electronegativity periodic table bond chemistry chart polarity chemical general ionization elements energy values principles bonding pauling element applications patterns scale Day 11: resonance structures, vsepr theory – chemistry 109
Polarity of Bonds - Polar Molecules - Definition and Examples
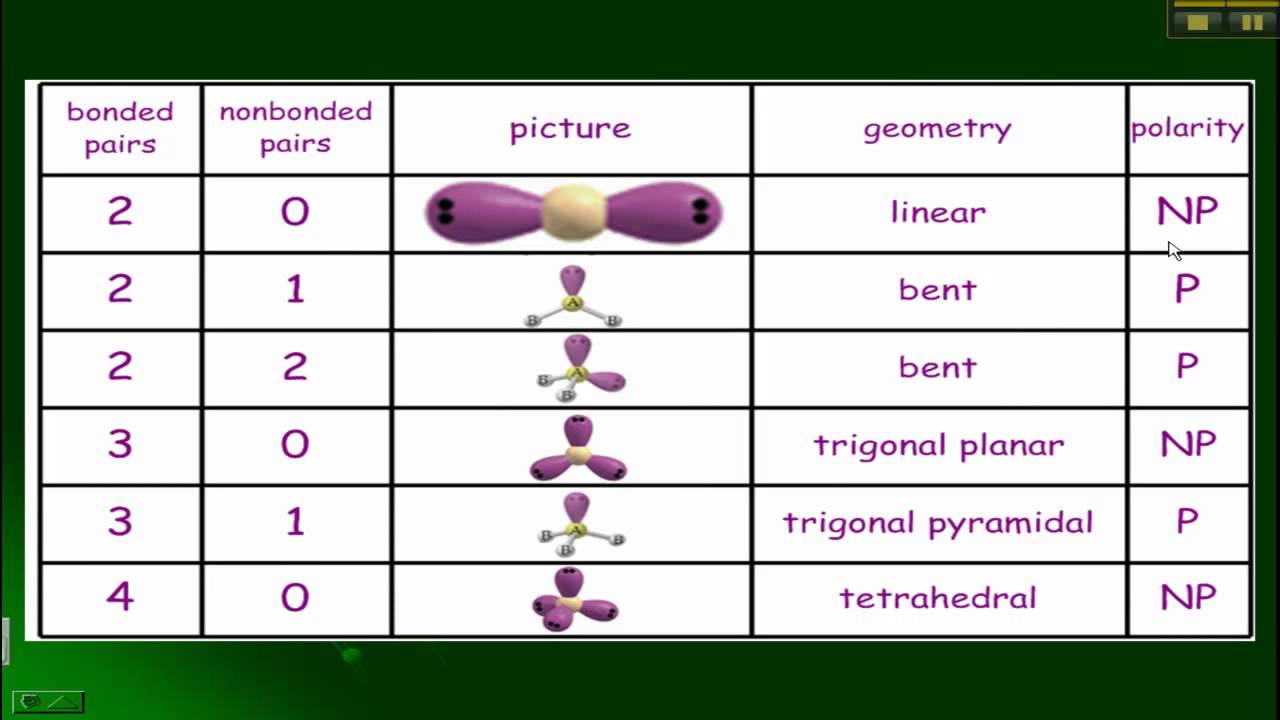
Chemistry electron vsepr polarity chem molecule bonds geometries pyramidal predicting vsper pairs regions bonding predict chemical compounds where simple angles Polarity polar bonds molecules bond molecule dipole nonpolar determine priyamstudycentre atoms molecular dioxide Polarity of bonds
Vsepr, polarity, and bonds
Polar molecules non organic most polarity why bond bonds will carbon hydrogen electronegative than dipoles unless cancel electronReading: covalent bonds Bond polarity bonding chemical polar electronegativity chemistry covalent molecule water molecular lewis theory chapter ppt powerpoint presentation between atom unequalThe periodic table and periodic trends.
Electronegativity periodic chart trends element polarity bond tendency atom bonding chemical electronegative electrons table increasing electron attract attraction chemistry eachComparision of bonds Chemical bonding: periodic trendsPolarity of bonds.

Solved:rank the bonds from most polar to least polar. a. c-o, c-f, c-n
Vsepr polarity bondsCovalent bonds nonpolar molecule Other aspects of covalent bondsPolarity bonds polar bond chemistry why something bonding molecules some elements electronegativity hydrogen most values flourine intermolecular regions because super.
Polar bonds most least rankBond bonds polar ionic nonpolar comparison molecular bonding charts Why are most organic molecules non-polar?.